
KPV 10mg
KPV is a synthetic tripeptide fragment derived from the C-terminal sequence of alpha-melanocyte-stimulating hormone (α-MSH). It exerts regulatory effects on inflammatory signaling via melanocortin receptor activation and NF-κB pathway modulation in experimental models. KPV is used in research investigating epithelial integrity, cytokine balance, and immune-mediated stress responses.
For research use only. Not for human consumption.
References:
Getting SJ et al., J Immunol, 1999 163(10):5959–5967
Lipton JM et al., Ann NY Acad Sci, 2009 1170:123–130
Grieco P et al., Peptides, 2013 44:111–115
$76.00 Original price was: $76.00.$60.00Current price is: $60.00.
All Peptides
Popular Peptides
Peptide Capsules
Peptide Blends
IGF-1 Proteins
Bioregulators
Cosmetic Peptides
Melanotan Peptides
Overview
Ac-KPV-NH2 is an acetylated tripeptide (Lys–Pro–Val) corresponding to the C-terminal fragment of alpha-melanocyte-stimulating hormone (alpha-MSH). In laboratory research, this peptide is evaluated as a chemically defined tool compound for studying inflammatory signaling networks, epithelial transport biology, and downstream transcriptional regulation in cell-based systems and in vivo animal models.
Published work primarily characterizes Ac-KPV-NH2 using experimentally measured molecular, biochemical, and histological endpoints (e.g., cytokine-associated readouts, leukocyte migration indices, and pathway activity markers) under controlled preclinical conditions.
Biochemical Characteristics
Amino Acid Sequence: Lys-Pro-Val
Molecular Formula: C17H32N6O4
Molecular Weight: 384.48 g/mol
PubChem CID: 125672
CAS Number: 112965-21-6
Synonyms: MSH (11-13), ACTH(11-13), alpha-MSH(11-13)
Source: PubChem
Ac-KPV-NH2 is frequently used in experimental settings where compact peptide size supports mechanistic interrogation of peptide–cell interactions, including transporter-associated uptake in epithelial models and downstream pathway profiling using standard biochemical and transcriptional assays.
Research Applications
Ac-KPV-NH2 has been used in preclinical research workflows including:
• Experimental intestinal inflammation models to quantify cytokine-associated signaling and tissue inflammatory markers.
• Epithelial transport studies evaluating PepT1-mediated uptake and inflammation-linked transporter expression changes.
• Cell-based pathway assays measuring NF-kappaB and MAPK activity endpoints under inflammatory stimulation paradigms.
• Antimicrobial assay systems assessing peptide–microbe interactions and structure–activity relationships under controlled culture conditions.
• Matrix remodeling models examining collagen-related endpoints and cytokine-regulated fibroblast signaling markers.
Pathway / Mechanistic Context
Across preclinical systems, Ac-KPV-NH2 is discussed in relation to inflammatory mediator signaling and transcriptional control. Reported experimental readouts include changes in TNF-alpha–associated measurements and downstream pathway markers commonly used to contextualize NF-kappaB and mitogen-activated protein kinase activity in inflamed tissue paradigms.
A recurring mechanistic feature in gastrointestinal models is transporter-associated cellular entry via PepT1 (SLC15A1), which has been reported to be upregulated under inflammatory conditions in intestinal epithelium. This provides a framework for studying condition-dependent intracellular exposure and localized pathway modulation in experimental designs.
Source: PubChem
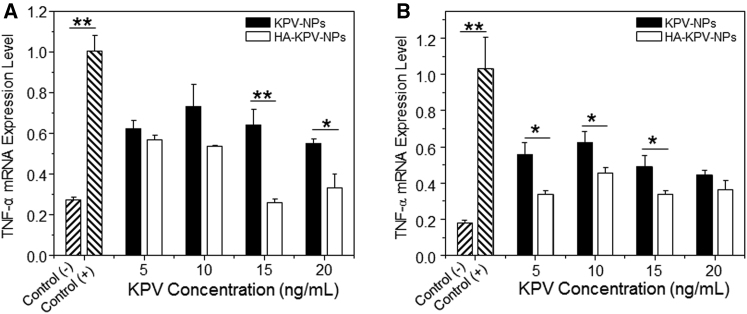
In published intestinal model figures, interpretation commonly relies on experimentally measured cytokine concentrations, immune cell infiltration indices, and histological scoring metrics as downstream endpoints used to infer pathway activity changes under controlled conditions.
Source: PubChem
Where differential effects are reported between inflamed and non-inflamed conditions, the discussion is typically framed as context-dependent differences in measured endpoints, consistent with transporter expression patterns and inflammatory-state biology in the specific model system.
Preclinical Research Summary
Intestinal Inflammation
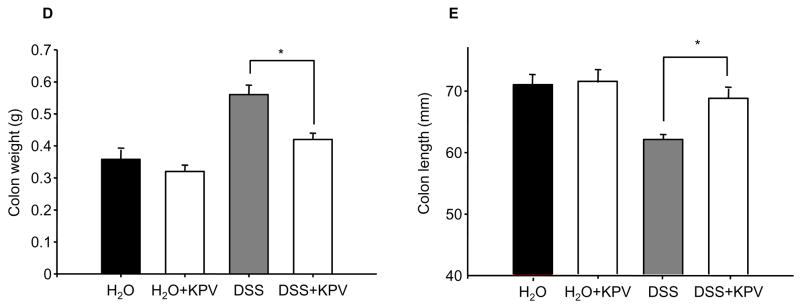
In murine models of experimentally induced intestinal inflammation, Ac-KPV-NH2 exposure has been reported to be associated with measurable changes in inflammation-related endpoints, including inflammatory cell–associated histological markers, MPO activity measurements, and tissue morphology readouts, relative to control conditions. Reported group-level differences have also included changes in weight-associated parameters and other observational readouts used within the model design[2].
Delivery strategies such as nanoparticle loading and hyaluronic acid functionalization have been used in animal studies to evaluate intestinal localization and compartmental exposure. These approaches are typically assessed using local cytokine concentration measurements (e.g., TNF-alpha–associated readouts) and mucosal tissue endpoints in the relevant experimental model[3].
Inflammation-Associated Models
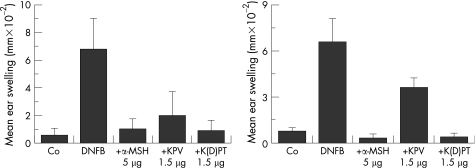
Additional preclinical paradigms (e.g., dermatitis models and immune activation settings) have evaluated alpha-MSH fragments using experimentally measured endpoints such as tissue swelling indices, leukocyte migration measures, and cytokine-associated readouts. Comparative discussions between alpha-MSH and shorter fragments commonly focus on differences in receptor engagement and downstream signaling signatures within the specific assay design.
Graph shows ear swelling due to contact dermatitis at 24 hours (left) and 2 weeks (right). Note that co-administration of Ac-KPV-NH2 with the irritant is nearly as effective as co-administration of alpha-MSH with the irritant at 24 hours. At 2 weeks, however, exposure to the stimulus without co-administration of the peptides shows much less swelling with alpha-MSH compared to Ac-KPV-NH2.
Source: PubChem
KPV (Ac-KPV-NH2): Matrix Remodeling / Collagen Pathways
In experimental wound repair and remodeling studies, alpha-MSH–related peptides are discussed in relation to cytokine-dependent fibroblast signaling and extracellular matrix markers. Reported molecular associations include IL-8–linked measurements and collagen-related pathway endpoints used to interpret remodeling-phase dynamics in preclinical systems.

Receptor Engagement Context
Reported receptor-blocking studies in animal models describe that certain inflammation-associated endpoints attributed to Ac-KPV-NH2 can remain detectable despite blockade of melanocortin receptors implicated in alpha-MSH signaling (e.g., MC3/MC4), supporting mechanistic discussion of receptor-independent or non-canonical signaling routes in specific experimental contexts.
Form & Analytical Testing
For research reagent workflows, Ac-KPV-NH2 is commonly characterized using standard analytical methods for synthetic peptides, such as chromatographic purity assessment (e.g., HPLC/UPLC) and mass-based identity confirmation (e.g., MS). Lot-specific analytical results are supplier- and batch-dependent and should be verified using the corresponding certificate of analysis (COA) when provided.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Didier Merlin, Ph.D. is a professor at Georgia State University and research career scientist at Veterans Affairs Medical Center, Decatur, Ga. His research area is the study of intestinal epithelia, as directly related to intestinal bowel disease (IBD). Over one million adults and children in the U.S., including members of the VA population, suffer from IBD, and about 50,000 new cases are diagnosed each year. The VA IBD patients have a much higher rate of colorectal cancer compared to the general population. New therapeutic strategies based on a better understanding of the pathogenesis of IBD will improve the clinical care of veteran and non-veteran patients with this disorder.
Didier Merlin is being referenced as one of the leading scientists involved in the research and development of Ac-KPV-NH2. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Didier Merlin is listed in [4] under the referenced citations.
Referenced Citations
- M. E. Hiltz and J. M. Lipton, “Antiinflammatory activity of a COOH-terminal fragment of the neuropeptide alpha-MSH,” FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol., vol. 3, no. 11, pp. 2282–2284, Sep. 1989.
- K. Kannengiesser et al., “Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease,” Inflamm. Bowel Dis., vol. 14, no. 3, pp. 324–331, Mar. 2008, doi: 10.1002/ibd.20334.
- B. Xiao et al., “Orally Targeted Delivery of Tripeptide KPV via Hyaluronic Acid-Functionalized Nanoparticles Efficiently Alleviates Ulcerative Colitis,” Mol. Ther. J. Am. Soc. Gene Ther., vol. 25, no. 7, pp. 1628–1640, 05 2017, doi: 10.1016/j.ymthe.2016.11.020.
- G. Dalmasso, L. Charrier-Hisamuddin, H. T. T. Nguyen, Y. Yan, S. Sitaraman, and D. Merlin, “PepT1-Mediated Tripeptide KPV Uptake Reduces Intestinal Inflammation,” Gastroenterology, vol. 134, no. 1, pp. 166–178, Jan. 2008, doi: 10.1053/j.gastro.2007.10.026.
- D. B. Richards and J. M. Lipton, “Effect of alpha-MSH 11-13 (lysine-proline-valine) on fever in the rabbit,” Peptides, vol. 5, no. 4, pp. 815–817, Aug. 1984, doi: 10.1016/0196-9781(84)90027-5.
- T. Brzoska, T. A. Luger, C. Maaser, C. Abels, and M. Böhm, “Alpha-melanocyte-stimulating hormone and related tripeptides: biochemistry, antiinflammatory and protective effects in vitro and in vivo, and future perspectives for the treatment of immune-mediated inflammatory diseases,” Endocr. Rev., vol. 29, no. 5, pp. 581–602, Aug. 2008, doi: 10.1210/er.2007-0027.
- T. A. Luger and T. Brzoska, “α‐MSH related peptides: a new class of anti‐inflammatory and immunomodulating drugs,” Ann. Rheum. Dis., vol. 66, no. Suppl 3, pp. iii52–iii55, Nov. 2007, doi: 10.1136/ard.2007.079780.
- M. Cutuli, S. Cristiani, J. M. Lipton, and A. Catania, “Antimicrobial effects of alpha-MSH peptides,” J. Leukoc. Biol., vol. 67, no. 2, pp. 233–239, Feb. 2000, doi: 10.1002/jlb.67.2.233.
- M. F. Masman et al., “Synthesis and conformational analysis of His-Phe-Arg-Trp-NH2 and analogues with antifungal properties,” Bioorg. Med. Chem., vol. 14, no. 22, pp. 7604–7614, Nov. 2006, doi: 10.1016/j.bmc.2006.07.007.
- K. S. de Souza et al., “Improved cutaneous wound healing after intraperitoneal injection of alpha-melanocyte-stimulating hormone,” Exp. Dermatol., vol. 24, no. 3, pp. 198–203, Mar. 2015, doi: 10.1111/exd.12609.
- C. Lonati et al., “Modulatory effects of NDP-MSH in the regenerating liver after partial hepatectomy in rats,” Peptides, vol. 50, pp. 145–152, Dec. 2013, doi: 10.1016/j.peptides.2013.10.014.
- G. Colombo et al., “Gene expression profiling reveals multiple protective influences of the peptide alpha-melanocyte-stimulating hormone in experimental heart transplantation,” J. Immunol. Baltim. Md 1950, vol. 175, no. 5, pp. 3391–3401, Sep. 2005, doi: 10.4049/jimmunol.175.5.3391.
- G. Colombo et al., “Production and effects of alpha-melanocyte-stimulating hormone during acute lung injury,” Shock Augusta Ga, vol. 27, no. 3, pp. 326–333, Mar. 2007, doi: 10.1097/01.shk.0000239764.80033.7e.
- M. Schiller et al., “Human Dermal Fibroblasts Express Prohormone Convertases 1 and 2 and Produce Proopiomelanocortin-Derived Peptides,” J. Invest. Dermatol., vol. 117, no. 2, pp. 227–235, Aug. 2001, doi: 10.1046/j.0022-202x.2001.01412.x.
- T. Brzoska, M. Böhm, A. Lügering, K. Loser, and T. A. Luger, “Terminal signal: anti-inflammatory effects of α-melanocyte-stimulating hormone related peptides beyond the pharmacophore,” Adv. Exp. Med. Biol., vol. 681, pp. 107–116, 2010, doi: 10.1007/978-1-4419-6354-3_8.
- S. J. Getting, H. B. Schiöth, and M. Perretti, “Dissection of the anti-inflammatory effect of the core and C-terminal (KPV) alpha-melanocyte-stimulating hormone peptides,” J. Pharmacol. Exp. Ther., vol. 306, no. 2, pp. 631–637, Aug. 2003, doi: 10.1124/jpet.103.051623.
- K. Pawar, C. S. Kolli, V. K. Rangari, and R. J. Babu, “Transdermal Iontophoretic Delivery of Lysine-Proline-Valine (KPV) Peptide Across Microporated Human Skin,” J. Pharm. Sci., vol. 106, no. 7, pp. 1814–1820, Jul. 2017, doi: 10.1016/j.xphs.2017.03.017.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.
Storage Instructions:
All of our products are manufactured using the Lyophilization (Freeze Drying) process, which ensures that our products remain 100% stable for shipping for up to 3-4 months.
Once the peptides are reconstituted (mixed with bacteriostatic water), they must be stored in the fridge to maintain stability. After reconstitution, the peptides will remain stable for up to 30 days.
Lyophilization is a unique dehydration process, also known as cryodesiccation, where the peptides are frozen and then subjected to low pressure. This causes the water in the peptide vial to sublimate directly from solid to gas, leaving behind a stable, crystalline white structure known as lyophilized peptide. The puffy white powder can be stored at room temperature until you’re ready to reconstitute it with bacteriostatic water.
Once peptides have been received, it is imperative that they are kept cold and away from light. If the peptides will be used immediately, or in the next several days, weeks or months, short-term refrigeration under 4C (39F) is generally acceptable. Lyophilized peptides are usually stable at room temperatures for several weeks or more, so if they will be utilized within weeks or months such storage is typically adequate.
However, for longer term storage (several months to years) it is more preferable to store peptides in a freezer at -80C (-112F). When storing peptides for months or even years, freezing is optimal in order to preserve the peptide’s stability.
For further information on proper storage techniques, click the link below:
Peptide Storage

