
SLU-PP-332 10MG
SLU-PP-332 is a small-molecule research compound identified as a selective agonist of peroxisome proliferator-activated receptor delta (PPARδ). It enhances fatty acid oxidation, mitochondrial biogenesis, and metabolic efficiency in preclinical models by modulating PPARδ-dependent gene expression. SLU-PP-332 is utilized in metabolic and mitochondrial research examining lipid metabolism, endurance signaling, and energy regulation mechanisms.
References:
Narkar VA et al., Cell, 2008;134(3):405–415
Fan W et al., J Biol Chem, 2004;279(7):5241–5248
Billon C et al., Cell Metab, 2022;34(1):69–84
These capsules are provided exclusively as inert containers for the dry transport, storage, and handling of laboratory research materials. They are not dosage forms and are not intended for ingestion or administration. They must not be used as drug delivery systems, consumed, or administered in any form. For Research Use Only: Not for use in humans or animals. Not for diagnostic or therapeutic purposes.
$160.00 Original price was: $160.00.$140.00Current price is: $140.00.
All Peptides
Popular Peptides
Peptide Capsules
Peptide Blends
IGF-1 Proteins
Bioregulators
Cosmetic Peptides
Melanotan Peptides
SLU-PP-332: Overview
SLU-PP-332 is a low-molecular-weight synthetic small molecule developed as a selective agonist of estrogen-related orphan receptors (ERRs), with primary activity at the ERRα and ERRγ isoforms. Estrogen-related receptors are ligand-activated transcription factors that localize to the nucleus and regulate gene expression programs associated with cellular energy metabolism, mitochondrial biogenesis, and oxidative phosphorylation.
SLU-PP-332 has been characterized exclusively in preclinical research systems, including in-vitro cellular models and in-vivo animal studies, where it is used as a molecular tool to interrogate ERR-dependent transcriptional networks and mitochondrial regulatory pathways.
SLU-PP-332: Biochemical Characteristics
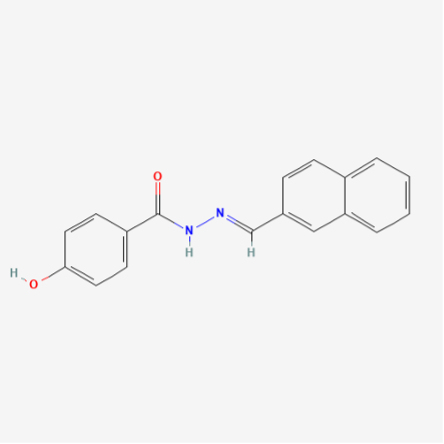
Source: PubChem
Chemical Formula: C18H14N2O2
Molecular Weight: 290.3 g/mol
PubChem CID: 5338394
CAS No.: 303760-60-3
Synonyms: 4-Hydroxy-N’-(naphthalen-2-ylmethylene) benzohydrazide
SLU-PP-332 is chemically stable under standard laboratory conditions and has demonstrated intracellular nuclear accessibility in experimental systems, enabling direct modulation of ERR-mediated transcriptional activity.
SLU-PP-332: Research Applications
SLU-PP-332 is supplied strictly for laboratory research use and has been employed in controlled experimental models to investigate:
- ERRα- and ERRγ-dependent transcriptional regulation
- Mitochondrial biogenesis and ultrastructural remodeling
- Oxidative phosphorylation and fatty acid oxidation pathways
- Cellular energy homeostasis and metabolic gene networks
- Autophagy-associated transcriptional signaling
All applications are limited to non-clinical, non-diagnostic experimental systems.
SLU-PP-332: Pathway / Mechanistic Context
Estrogen-related receptors function as constitutively active transcription factors that bind ERR response elements within promoter regions of genes governing mitochondrial respiration, lipid metabolism, and oxidative stress regulation. SLU-PP-332 has been shown in preclinical models to enhance ERRα- and ERRγ-mediated transcriptional output.
Downstream effects observed in experimental systems include increased expression of genes associated with electron transport chain assembly, mitochondrial fatty acid uptake, and transcription factor EB (TFEB)-regulated autophagy pathways. These effects position SLU-PP-332 as a mechanistic probe for studying mitochondrial quality control and energy regulation at the transcriptional level.
SLU-PP-332: Preclinical Research Summary
Preclinical investigations utilizing rodent and cellular models have demonstrated that SLU-PP-332 modulates ERR-dependent gene networks governing mitochondrial density, substrate utilization, and oxidative capacity. These studies report alterations in mitochondrial morphology, respiratory efficiency, and lipid oxidation under controlled experimental conditions.
Additional research has examined SLU-PP-332 in models of metabolic stress, cardiac energetic demand, renal mitochondrial aging, and neuronal mitochondrial maintenance, providing insight into conserved ERR-regulated bioenergetic pathways.
SLU-PP-332: Form & Analytical Testing
SLU-PP-332 is supplied as a research-grade small molecule compound. Each lot is produced under controlled conditions and verified using standard analytical techniques, including chromatographic purity assessment and mass-based identity confirmation.
Analytical data are provided solely to support experimental reproducibility.
About The Author
The above literature was researched, edited, and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Dr. Hamid Nasri, M.D., is a Clinical Nephropathologist and Nephrologist affiliated with the Department of Natural Sciences at The University of Georgia in Tbilisi, Georgia. He is also a retired Professor of Nephrology as of February 2024.
Dr. Hamid Nasri, M.D., is referenced as one of the leading scientists involved in the research and development of SLU-PP-332. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr.Hamid Nasri, M.D., is listed in [1] under the referenced citations.
Resources
- H. Nasri, “New hopes on ‘SLU-PP-332’ as an effective agent for weight loss with indirect kidney protection efficacy; a nephrology point of view,” J Ren Endocrinol, vol. 10, no. 1, Art. no. 1, Jan. 2024, doi: 10.34172/jre.2024.25143.
- J.-S. Wattez et al., “Loss of skeletal muscle estrogen-related receptors leads to severe exercise intolerance,” Mol Metab, vol. 68, p. 101670, Jan. 2023, doi: 10.1016/j.molmet.2023.101670.
- F. Xin, L. M. Smith, M. Susiarjo, M. S. Bartolomei, and K. J. Jepsen, “Endocrine-disrupting chemicals, epigenetics, and skeletal system dysfunction: exploration of links using bisphenol A as a model system,” Environ Epigenet, vol. 4, no. 2, p. dvy002, Apr. 2018, doi: 10.1093/eep/dvy002.
- S. N. Fox et al., “Estrogen-related receptor gamma regulates mitochondrial and synaptic genes and modulates vulnerability to synucleinopathy,” npj Parkinsons Dis., vol. 8, no. 1, pp. 1–19, Aug. 2022, doi: 10.1038/s41531-022-00369-w.
- C. Billon et al., “Synthetic ERRα/β/γ Agonist Induces an ERRα-Dependent Acute Aerobic Exercise Response and Enhances Exercise Capacity,” ACS Chem. Biol., vol. 18, no. 4, pp. 756–771, Apr. 2023, doi: 10.1021/acschembio.2c00720.
- J. A. Hawley, M. J. Joyner, and D. J. Green, “Mimicking exercise: what matters most and where to next?,” J Physiol, vol. 599, no. 3, pp. 791–802, Feb. 2021, doi: 10.1113/JP278761.
- “Mimicking exercise with a pill,” American Chemical Society. Accessed: Jan. 23, 2025. [Online]. Available: https://www.acs.org/pressroom/presspacs/2024/march/mimicking-exercise-with-a-pill.html
- “Weight loss: Exercise-mimicking drug may reduce fat, improve insulin.” Accessed: Jan. 23, 2025. [Online]. Available: https://www.medicalnewstoday.com/articles/new-drug-may-help-lose-weight-reduce-fat-by-mimicking-exercise
- P.-M. Badin et al., “Exercise-like effects by Estrogen-related receptor-gamma in muscle do not prevent insulin resistance in db/db mice,” Sci Rep, vol. 6, no. 1, p. 26442, May 2016, doi: 10.1038/srep26442.
- W. Xu et al., “Novel pan-ERR agonists ameliorate heart failure through enhancing cardiac fatty acid metabolism and mitochondrial function,” Circulation, vol. 149, no. 3, pp. 227–250, Jan. 2024, doi: 10.1161/CIRCULATIONAHA.123.066542.
- M. Losby et al., “The Estrogen Receptor-Related Orphan Receptors Regulate Autophagy through TFEB,” Mol Pharmacol, vol. 106, no. 4, pp. 164–172, Sep. 2024, doi: 10.1124/molpharm.124.000889.
- X. X. Wang et al., “Estrogen-Related Receptor Agonism Reverses Mitochondrial Dysfunction and Inflammation in the Aging Kidney,” Am J Pathol, vol. 193, no. 12, pp. 1969–1987, Dec. 2023, doi: 10.1016/j.ajpath.2023.07.008.
- S. Miwa, S. Kashyap, E. Chini, and T. von Zglinicki, “Mitochondrial dysfunction in cell senescence and aging,” J Clin Invest, vol. 132, no. 13, p. e158447, Jul. 2022, doi: 10.1172/JCI158447.
- E. Schoepke et al., “A Selective ERRα/γ Inverse Agonist, SLU-PP-1072, Inhibits the Warburg Effect and Induces Apoptosis in Prostate Cancer Cells,” ACS Chem Biol, vol. 15, no. 9, pp. 2338–2345, Sep. 2020, doi: 10.1021/acschembio.0c00670.
- A. Rodríguez, “Not committed to fail: novel approach improves heart failure outcomes in animal model,” Baylor College of Medicine Blog Network. Accessed: Jan. 23, 2025. [Online]
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
RUO Disclaimer
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.

